Drug–Drug Interactions: A Pharmacological and Pharmaceutical Perspective on Formulation and Safety
DOI:
https://doi.org/10.64474/3107-6726.Vol2.Issue1.4Keywords:
- Drug–drug interactions, pharmacokinetics, pharmacodynamics, polypharmacy, formulation strategies, safety, predictive modeling
Abstract
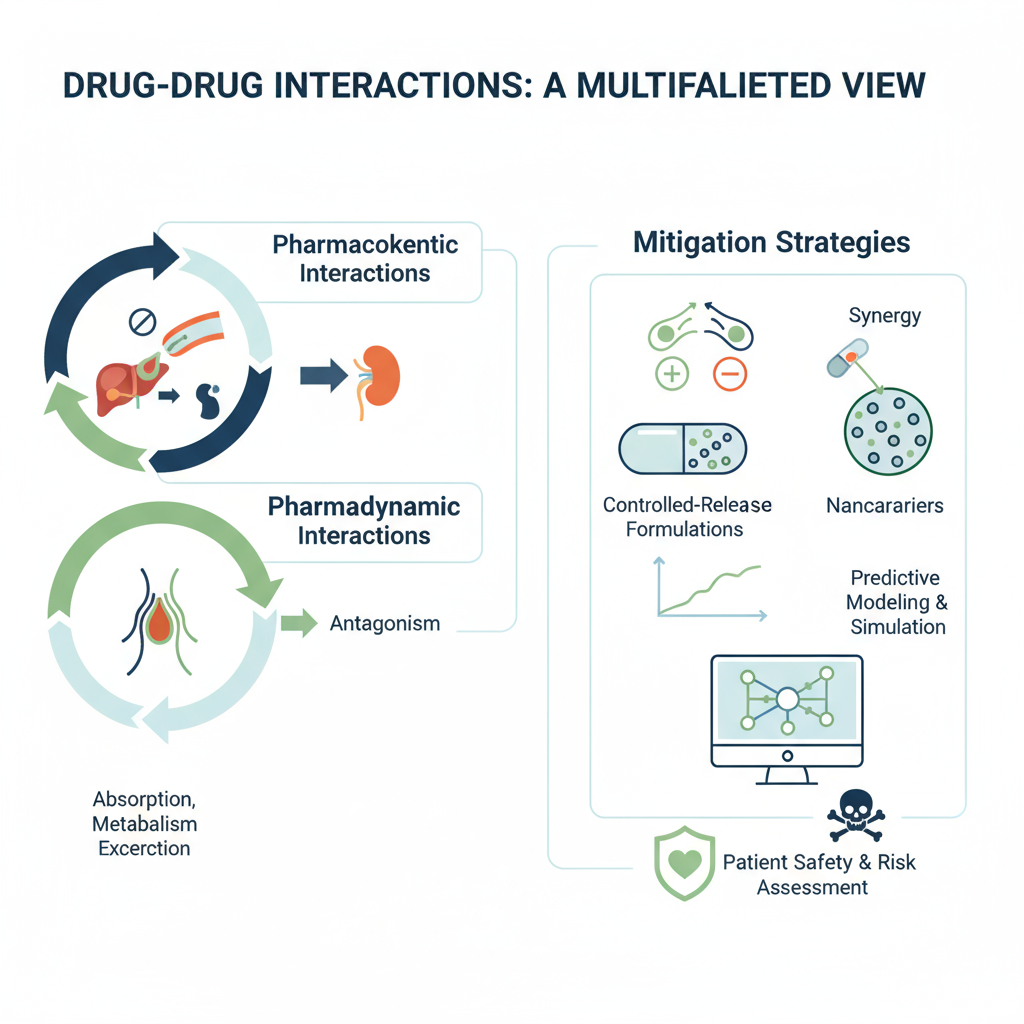
Drug–drug interactions (DDIs) are a critical consideration in clinical pharmacology and pharmaceutical development, affecting therapeutic efficacy and patient safety. DDIs may result from pharmacokinetic mechanisms (absorption, distribution, metabolism, elimination) or pharmacodynamic mechanisms (synergistic, antagonistic, or additive effects). This review explores the pharmacological basis of DDIs, their implications in formulation design, and strategies to predict, prevent, and manage adverse interactions. Emphasis is placed on emerging tools, including computational modeling, in vitro and in vivo assays, and regulatory considerations, highlighting the integration of pharmaceutical science with clinical pharmacology for safe drug administration.